
N3 Lewis Structure How to Draw the Lewis Structure for N3 YouTube
The molecular geometry about each N is trigonal pyramidal. The number of hybrid orbitals used by the central atom is the same as the number of electron pairs around the central atom. Hybridization Using d Orbitals. Hybridization is not restricted to the ns and np atomic orbitals. The bonding in compounds with central atoms in the period 3 and.
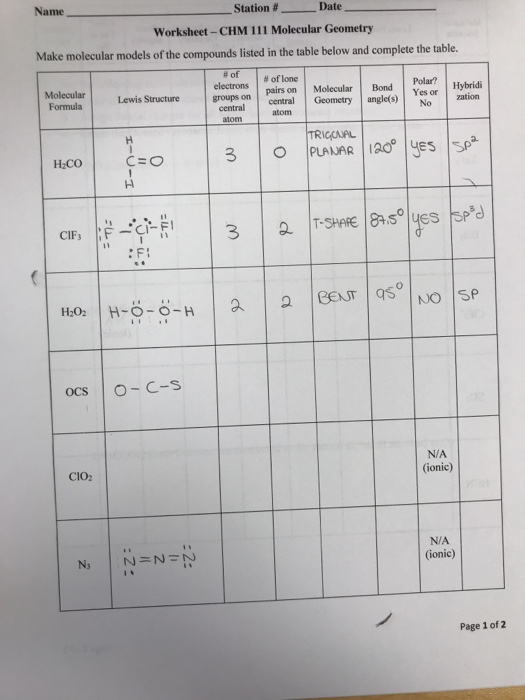
Solved Make molecular models of the compounds listed in the
Describe the molecular geometry of N 3 −. Molecular geometry: There are five electron-pair geometries: linear, trigonal planar, octahedral, tetrahedral, and trigonal bipyramidal.

CH3NH2 Lewis Structure, Molecular Geometry, Hybridization, and Polarity
In the N 3- Lewis structure, there are two double bonds around the nitrogen atom, with two other nitrogen atoms attached to it, and on the left and right nitrogen atoms, there are two lone pairs. Also, there is a negative (-1) charge on the left and right nitrogen atoms, and a positive (+1) charge on the center nitrogen atom.

Hybridization, Molecular Geometry and Bond Angles without/with lone
Geometry of Molecules. Molecular geometry, also known as the molecular structure, is the three-dimensional structure or arrangement of atoms in a molecule. Understanding the molecular structure of a compound can help determine the polarity, reactivity, phase of matter, color, magnetism, as well as the biological activity.
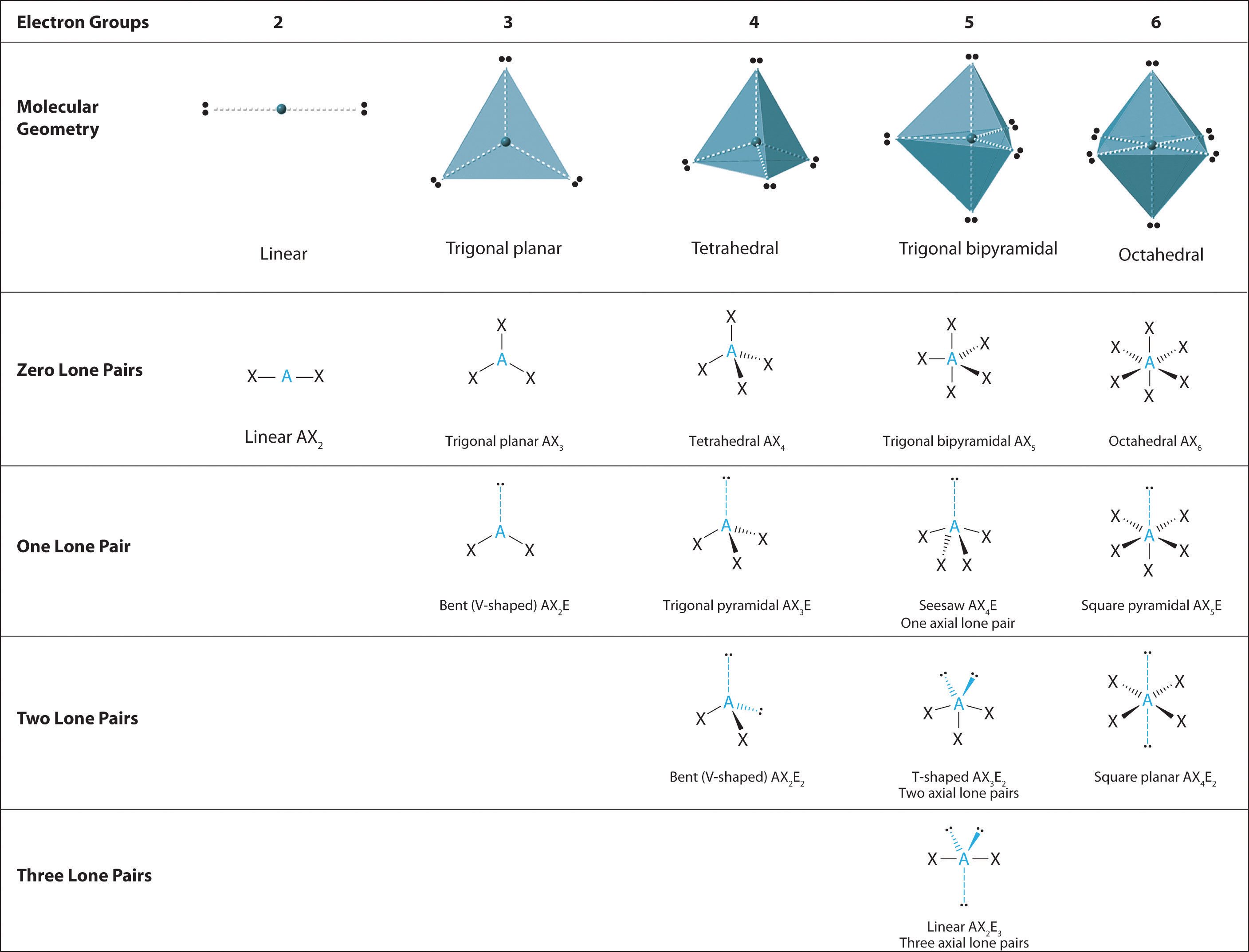
1.3 VSPER Theory The Effect of Lone Pairs Chemistry LibreTexts
1. N3- Lewis Structure: Here's a step-by-step guide on drawing the N3 - Lewis structure. Step 1: draw sketch • To begin, count the total amount of valence electrons. Nitrogen is in group 15 of the periodic table. As a result of this, nitrogen has five valence electrons. Because N 3- contains three nitrogen atoms,

The electron pair geometry and shape around C2 and N3 in the molecule
Azide. The azide anion. In chemistry, azide ( / ˈeɪzaɪd /, AY-zyd) is a linear, polyatomic anion with the formula N− 3 and structure −N=N+=N−. It is the conjugate base of hydrazoic acid HN3. Organic azides are organic compounds with the formula RN3, containing the azide functional group. [1] The dominant application of azides is as a.

Nitride Ion
The geometry of BCl3 BCl 3 is also given in Figure 7.2: it is trigonal planar, with all four atoms lying in the same plane, and all Cl−B−Cl Cl − B − Cl bond angles equal to 120o 120 o. The three Cl Cl atoms form an equilateral triangle. The Boron atom has only three pairs of valence shell electrons in BCl3 BCl 3.
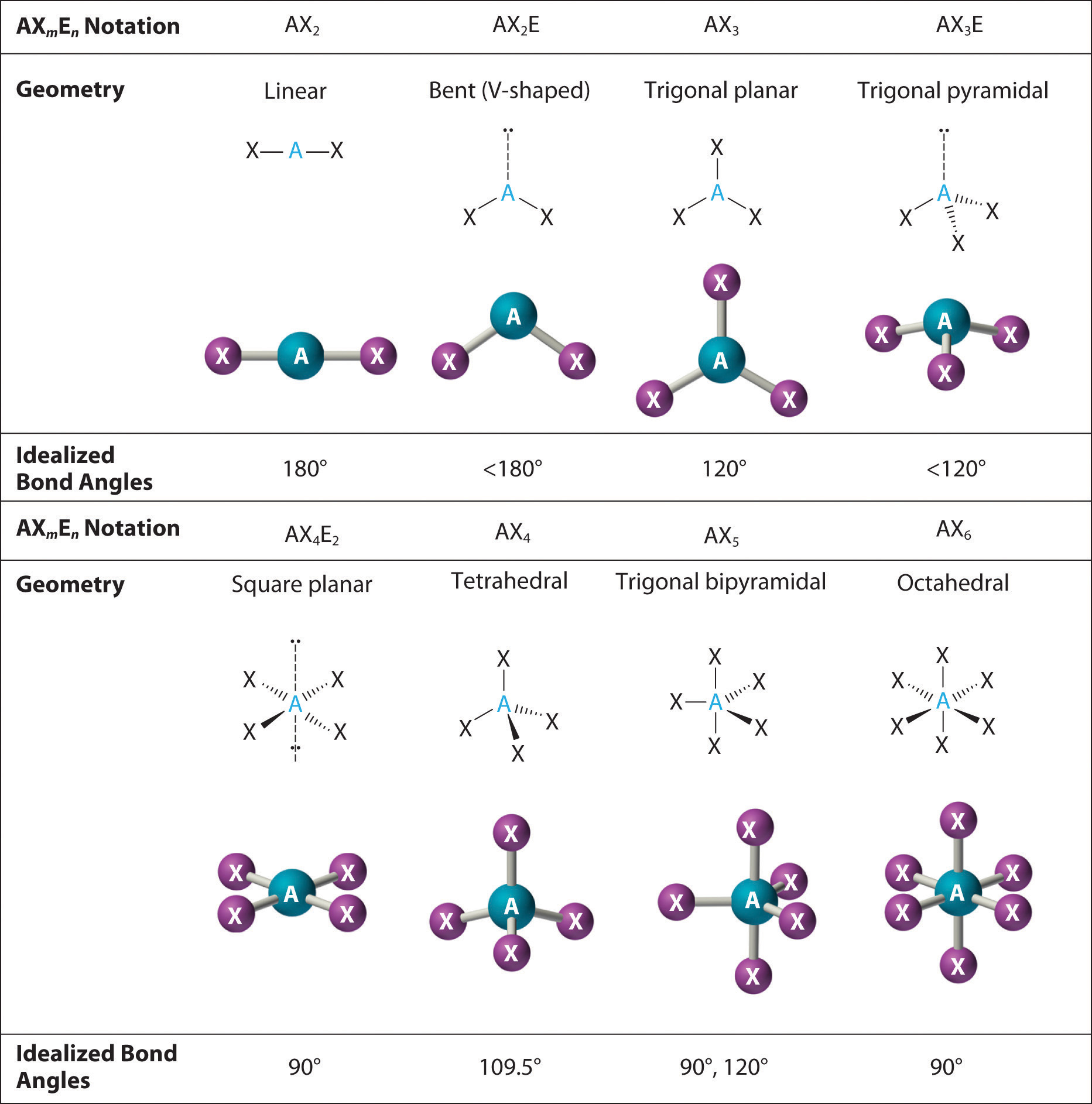
Predicting the Geometry of Molecules and Polyatomic Ions
I quickly take you through how to draw the Lewis Structure of N3- (Azide Ion) . I also go over hybridization, shape and bond angles.

N3 (Azide Ion) Molecular Geometry, Bond Angles & Electron Geometry
In the Lewis Structure for N3- you'll need to place a double bonds between the Nitrogen atoms to achieve full outer shells on all atoms while only using the valence electrons available for the molecule. For the N3- Lewis structure, calculate the total number of valence electrons for the N3- molecule.

Top N3 Molecular Geometry Pics GM
Description Azide anion is a pseudohalide anion. It has a role as a mitochondrial respiratory-chain inhibitor. It is a conjugate base of a hydrogen azide. ChEBI Organic or inorganic compounds that contain the -N3 group. Medical Subject Headings (MeSH) 1 Structures 1.1 2D Structure Structure Search Get Image Download Coordinates

AsF3 Molecular Geometry Science Education and Tutorials
D With two nuclei around the central atom and one lone pair of electrons, the molecular geometry of SnCl 2 is bent, like SO 2, but with a Cl-Sn-Cl bond angle of 95°. The molecular geometry can be described as a trigonal planar arrangement with one vertex missing. Exercise. Predict the molecular geometry of each molecule. SO 3; XeF 4.
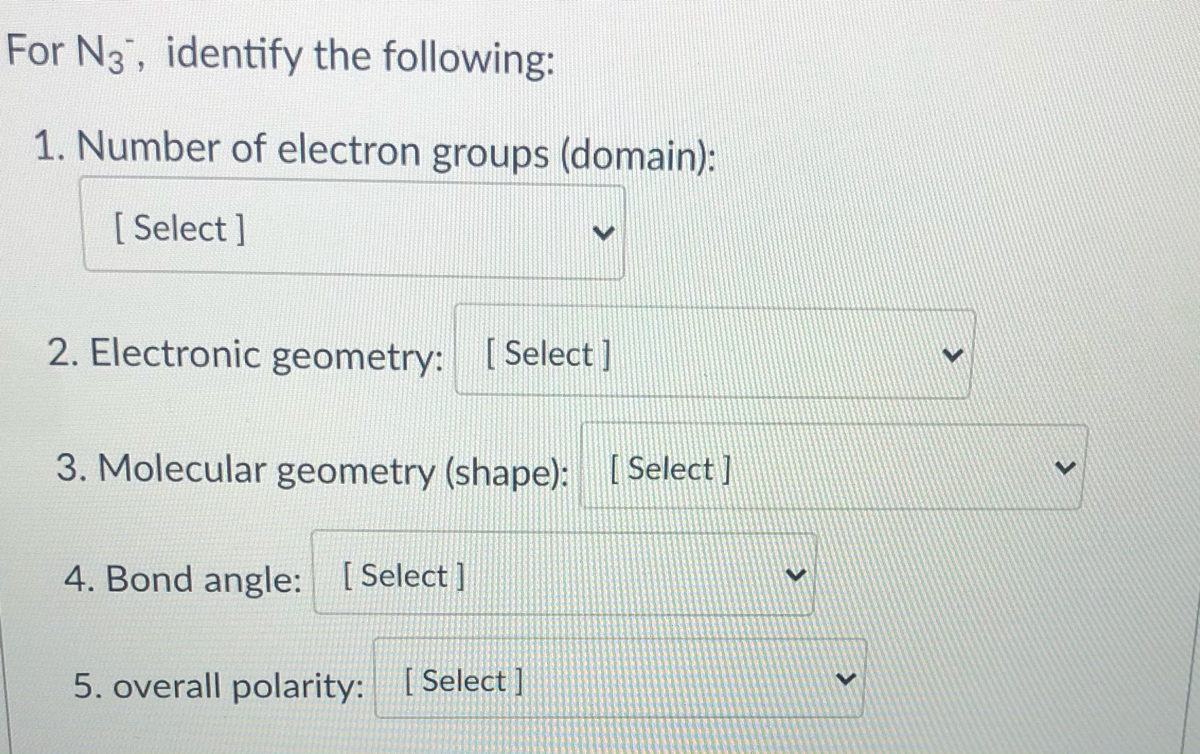
Answered For N3, identify the following 1.… bartleby
21K views 3 years ago An explanation of the molecular geometry for the N3 - ion (Azide Ion) including a description of the N3 - bond angles. The electron geometry for the Azide Ion is.

Answered Give the electrondomain and molecular… bartleby
Azide [N3]- ion Lewis structure, molecular geometry or shape, resonance structure, polar or non-polar, hybridization, bond angle N 3- is the chemical formula for the azide ion, also known as hydrazoate. It is an anion composed of three nitrogen (N) atoms. It is the conjugate base of hydrazoic acid/ hydrogen azide (HN 3 ).

Lone Pair of Electrons
Nitrogen is a group VA element in the periodic table and contains five electrons in its last shell. To find out total valence electrons given by a particular element, you should multiply number of electrons of the valance shell by the number of atoms of that element in respective molecule. valence electrons given by nitrogen atoms = 5*3 = 15

11333.jpg
D With two nuclei around the central atom and one lone pair of electrons, the molecular geometry of SnCl 2 is bent, like SO 2, but with a Cl-Sn-Cl bond angle of 95°. The molecular geometry can be described as a trigonal planar arrangement with one vertex missing. Exercise. Predict the molecular geometry of each molecule. SO 3; XeF 4.

Is NO3 Polar or Nonpolar? Techiescientist
Chemistry Chemistry questions and answers 4. Using the Lewis dot structure, predict the molecular geometry of the following and state the hybridization and bond angle of the central atom. a. N3- b. NO3- c. BF4- d. CH4 e. C2H2 This problem has been solved!